Osimertinib (Tagrisso): Latest Approval Updates for Early-Stage Lung Cancer — What Patients and Families Need to Know
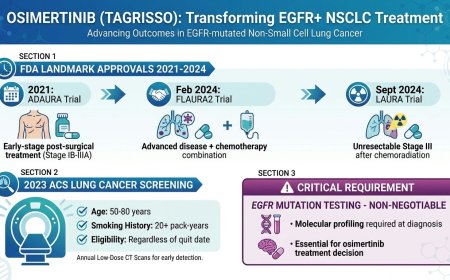
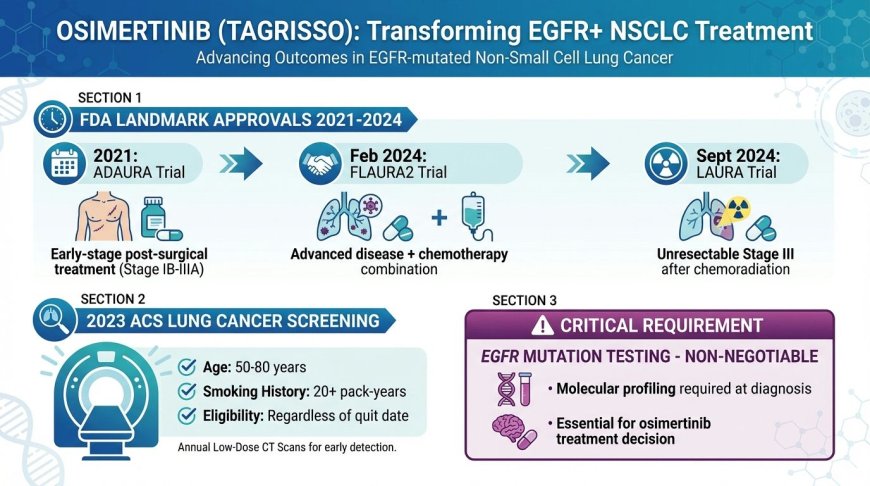
Osimertinib (Tagrisso) has become a transformative targeted therapy for EGFR-mutated non-small cell lung cancer. Between 2021 and 2024, the FDA granted landmark approvals covering early-stage post-surgical treatment (ADAURA trial, 2021), unresectable Stage III disease following chemoradiation (LAURA trial, September 2024), and advanced disease in combination with chemotherapy (FLAURA2 trial, February 2024). The 2023 ACS lung cancer screening guidelines now recommend annual low-dose CT scans for adults aged 50–80 with a 20+ pack-year smoking history, regardless of when they quit. Eligibility for osimertinib requires confirmed EGFR mutation testing — making molecular profiling at diagnosis absolutely non-negotiable for every NSCLC patient.
At a Glance
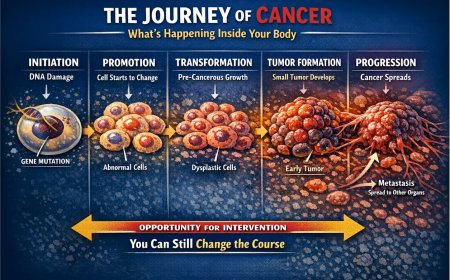
Lung cancer remains one of the most feared diagnoses a person can receive. But the landscape of treatment is changing — rapidly, and for the better. Osimertinib, sold under the brand name Tagrisso and developed by AstraZeneca, has become a cornerstone of targeted therapy for a specific and important subset of lung cancer. A series of landmark FDA approvals between 2021 and 2024 have expanded its role from advanced disease all the way back to early-stage settings, offering new hope to patients at every point along the disease journey. This article explains what these approvals mean, who qualifies, and what questions to ask your oncologist.
What Is Osimertinib (Tagrisso) and Why Does It Matter?
Osimertinib is a third-generation, irreversible EGFR tyrosine kinase inhibitor (EGFR-TKI). In plain language: it is an oral targeted therapy — a once-daily tablet — designed to block a specific protein called the Epidermal Growth Factor Receptor (EGFR) that drives the growth of certain lung cancer cells.
Not all lung cancers are the same. Non-small cell lung cancer (NSCLC) accounts for approximately 80–85% of all lung cancer diagnoses. Among patients with NSCLC, a subset carries specific genetic mutations in the EGFR gene — specifically what are known as exon 19 deletions or exon 21 L858R mutations. These mutations make the cancer particularly responsive to EGFR-targeted therapies like osimertinib.
Since its first accelerated approval in 2015 and subsequent regular approvals, Tagrisso has been used to treat nearly 800,000 patients worldwide across its approved indications. It is now approved as monotherapy in more than 100 countries, including the United States, European Union, China, and Japan.
Understanding the Approvals: A Clear Timeline
It is important to understand that osimertinib has received multiple distinct FDA approvals for different stages of lung cancer and different treatment settings. The most recent approvals are particularly significant for patients in earlier stages of the disease.
2021: Adjuvant Treatment After Surgery for Early-Stage NSCLC (ADAURA Trial)
The FDA approved osimertinib for adjuvant treatment — meaning treatment given after surgery aimed at cure — in adult patients with NSCLC whose tumors carry EGFR exon 19 deletions or exon 21 L858R mutations. This was a historic moment: it represented the first-ever FDA approval of a targeted adjuvant therapy for EGFR-mutated NSCLC.
The approval was supported by data from the Phase III ADAURA trial. Osimertinib reduced the risk of disease recurrence or death in patients with Stage II to IIIA disease by 83% compared to placebo. At 24 months, 90% of patients in the osimertinib group remained alive and disease-free, compared to 44% in the placebo group. Updated ADAURA results published in the Journal of Clinical Oncology in 2023 confirmed a significant overall survival benefit, cementing osimertinib as the standard of care in the post-surgical setting for eligible patients.
February 2024: Osimertinib Plus Chemotherapy for Advanced/Metastatic NSCLC (FLAURA2 Trial)
On February 16, 2024, the FDA approved osimertinib in combination with platinum-based chemotherapy for patients with locally advanced or metastatic NSCLC harboring EGFR exon 19 deletions or exon 21 L858R mutations. This was based on the Phase III FLAURA2 trial. Osimertinib plus chemotherapy reduced the risk of disease progression or death by 38% compared to osimertinib monotherapy alone. Median progression-free survival was 25.5 months with the combination versus 16.7 months with osimertinib monotherapy — an improvement of nearly nine months. This approval now gives oncologists two effective osimertinib-based options for the advanced setting, allowing more personalized treatment decisions.
September 2024: Osimertinib for Unresectable Stage III NSCLC After Chemoradiation (LAURA Trial)
On September 25, 2024, the FDA approved osimertinib for adult patients with locally advanced, unresectable (Stage III) NSCLC whose disease has not progressed during or following concurrent or sequential platinum-based chemoradiation therapy, and whose tumors have EGFR exon 19 deletions or exon 21 L858R mutations. This approval was based on the landmark LAURA trial, published in the New England Journal of Medicine in 2024 and presented at the ASCO Annual Meeting. Osimertinib demonstrated a median progression-free survival of 39.1 months compared to 5.6 months in the placebo arm — representing an 84% reduction in the risk of disease progression or death. This is now considered a potential new standard of care for this patient population, which previously had no approved targeted therapy option.
What These Approvals Mean in Practice
For the right patient, osimertinib now spans the entire spectrum of EGFR-mutated NSCLC treatment. Patients who have had surgery to remove early-stage lung cancer and carry EGFR mutations may now receive osimertinib as a post-surgical adjuvant therapy to reduce the risk of the cancer returning. Patients with unresectable Stage III disease who have completed standard chemoradiation without progression may now receive osimertinib as consolidation therapy — a targeted approach that dramatically prolongs the period before disease advances. Patients with advanced or metastatic disease now have two osimertinib-based first-line treatment options: osimertinib alone, or osimertinib combined with platinum-based chemotherapy.
This breadth of coverage across disease stages is unprecedented in EGFR-targeted therapy and represents a profound shift in how lung cancer is managed.
Who Is Eligible? The Critical Role of EGFR Testing
None of these approvals apply to all lung cancer patients. The eligibility requirement in every case is that the patient's tumor must carry a specific EGFR mutation — either an exon 19 deletion or an exon 21 L858R mutation — as detected by an FDA-approved test.
This is why molecular testing at the time of diagnosis is not optional — it is essential.
NCCN (National Comprehensive Cancer Network) guidelines, updated as recently as December 2025, now incorporate osimertinib across multiple stages of EGFR-mutated NSCLC. The American Society of Clinical Oncology (ASCO) similarly supports EGFR mutation testing as a mandatory step in the workup of newly diagnosed NSCLC.
If you or a loved one has been diagnosed with NSCLC, the first and most important question to ask is: "Has my tumor been tested for EGFR mutations and other molecular markers?" Without this information, critical treatment opportunities may be missed.
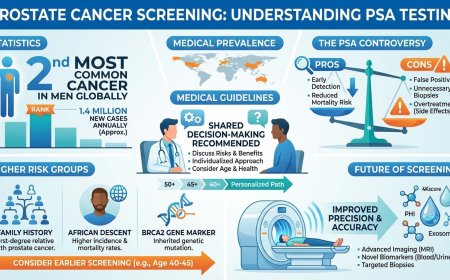
ACS Screening Guidelines: Who Should Be Tested for Lung Cancer?
Before targeted therapy can be considered, lung cancer must first be detected — ideally early, when treatment options are most effective. The American Cancer Society (ACS) released a significantly updated Lung Cancer Screening Guideline in November 2023, published in its flagship journal CA: A Cancer Journal for Clinicians. This update expanded the population recommended for annual screening.
The ACS now recommends annual low-dose CT (LDCT) scanning for asymptomatic adults aged 50 to 80 years who currently smoke or formerly smoked and have a smoking history of 20 or more pack-years. A pack-year is calculated as smoking one pack of cigarettes per day for one year; for example, a person who smoked one pack a day for 20 years, or two packs a day for 10 years, would meet the 20 pack-year threshold.
Critically, the 2023 guideline eliminated the previous requirement that former smokers must have quit within the past 15 years to qualify. Research now shows that lung cancer risk among former smokers does not decline as sharply with time since quitting as previously assumed — making the removal of the quit-year criterion an important expansion of screening eligibility. The guideline also expanded the eligible age range from the previous 55–74 years to the current 50–80 years, and lowered the smoking history threshold from 30 pack-years to 20 pack-years.
Before initiating screening, the ACS recommends that individuals engage in a shared decision-making discussion with a qualified healthcare professional to understand the purpose, process, potential benefits, limitations, and risks associated with LDCT screening.
For patients in India, lung cancer screening practices vary significantly by institution and region. Individuals with significant smoking histories — including those who use bidis, hookah, or other tobacco products — and those with occupational exposures to carcinogens such as asbestos or silica, or who live in high air-pollution urban environments, should proactively discuss lung cancer risk and screening options with their physician.
Real-Life Context: What Patients Often Don't Know
Consider the experience of patients like Anita, a 52-year-old non-smoker from Pune. She was diagnosed with Stage IIIA lung cancer after surgery. Her oncologist ordered molecular profiling and found an EGFR exon 19 deletion. She was started on adjuvant osimertinib — a once-daily oral tablet she takes at home. Two years later, she remains disease-free. Without molecular testing, she would not have known she was eligible.
Or consider Vikram, a 61-year-old retired engineer from Chennai. Diagnosed with unresectable Stage III NSCLC with an EGFR mutation, he completed chemoradiation. His oncologist, aware of the LAURA trial data and the September 2024 FDA approval, started him on osimertinib as consolidation therapy. His disease has not progressed.
These are not exceptional cases. They are the new standard — if the right tests are done and the right questions are asked.
Common Questions About Osimertinib
How is osimertinib taken?
Osimertinib is taken as an oral tablet, 80 mg once daily, with or without food. It does not require hospital visits for infusion like chemotherapy or many immunotherapy drugs.
What are the side effects?
Common side effects include rash, diarrhea, nail toxicity, musculoskeletal pain, and cough. A potentially serious but less common side effect is interstitial lung disease (ILD) or pneumonitis. Patients receiving osimertinib plus chemotherapy may experience additional side effects related to chemotherapy. All side effects should be discussed with and monitored by your oncologist.
How long is treatment continued?
Treatment continues until disease progression or unacceptable toxicity, according to prescribing guidelines. In the adjuvant setting, the ADAURA trial used three years of therapy.
Is osimertinib available in India?
Tagrisso is approved in more than 100 countries. Patients in India should consult their oncologist and confirm availability, as access and insurance coverage vary. Your oncologist can also advise on patient assistance programs offered by AstraZeneca.
What You Should Ask Your Oncologist Right Now
If you or a family member has been diagnosed with NSCLC, consider asking the following questions during your next consultation. Has comprehensive molecular testing been performed on my tumor, including for EGFR mutations? If I carry an EGFR exon 19 deletion or exon 21 L858R mutation, am I eligible for osimertinib? Which specific indication applies to my stage and treatment history — post-surgical adjuvant, post-chemoradiation consolidation, or first-line for advanced disease? Would the combination of osimertinib plus chemotherapy be appropriate in my case? Are there any ongoing clinical trials I should know about?
The quality of the conversation you have with your oncologist can directly influence your treatment options. Early testing opens doors. Delayed testing closes them.
Common Misconceptions That Can Delay Treatment
"My oncologist at the local hospital said chemotherapy is the standard." Chemotherapy may have been the standard before molecular profiling was routine. In 2025 and beyond, comprehensive molecular testing is itself the standard for NSCLC — as established by NCCN, ASCO, and international oncology guidelines.
"I'm not sure my insurance will cover molecular testing." Comprehensive genomic profiling for NSCLC is increasingly covered by major insurers. If there is uncertainty, your oncologist or hospital's financial counselor can help navigate this.
"I feel okay right now, so there's no urgency." The window for adjuvant therapy after surgery is time-sensitive. The decision about consolidation therapy after chemoradiation must also be made within a defined timeframe. Delays in these settings can mean forgoing a treatment that is available today.
The Bigger Picture: Targeted Therapy and the Future of Lung Cancer Care
The approvals of osimertinib across disease stages reflect a broader revolution in oncology — the movement away from one-size-fits-all chemotherapy toward therapies that match the unique molecular fingerprint of each patient's cancer. Osimertinib is the only targeted therapy to have demonstrated improved outcomes in Phase III clinical trials across early-stage disease (ADAURA), locally advanced unresectable disease (LAURA), and advanced/metastatic disease (FLAURA and FLAURA2). AstraZeneca continues to investigate osimertinib in the neoadjuvant (pre-surgical) setting through the NeoADAURA trial, with results anticipated in the near future.
This trajectory suggests that as testing becomes more routine and earlier, the number of patients who can benefit from targeted approaches will continue to grow.
Professional Support Options
If you or a loved one has received a lung cancer diagnosis, is seeking a second opinion on EGFR testing results, or wants guidance on whether osimertinib is appropriate for your specific case, do not navigate this alone. Speak with a qualified oncologist today.
Connect with experienced U.S.-based cancer specialists for a comprehensive second-opinion consultation. They will carefully review your case and help determine the most appropriate next steps for your individual health needs:
👉 https://myamericandoctor.com/our-doctors/
You may also choose to enroll in our upcoming concierge medical clinic in India, Global Concierge Doctors. We offer U.S.-style primary care with 24/7 access to India-based physicians for ongoing guidance on any health concern. When required, we coordinate referrals to trusted specialists in India and the U.S. for advanced evaluation and care.
Your health decisions today shape your life tomorrow.
Reader Poll
Has your oncologist discussed EGFR mutation testing with you or a family member?
- [ ] Yes, testing was done and results guided treatment
- [ ] Testing was mentioned but not yet completed
- [ ] No, I was not aware this testing existed
- [ ] I am a healthcare professional researching this topic
Share This Article
Know someone recently diagnosed with lung cancer who may not be aware of EGFR testing or the latest osimertinib approvals? Share this article. It could open a conversation that changes their treatment path.
Last Updated: 20th February 2026
Sources and References Consulted
The following authoritative sources were consulted in the preparation of this article:
-
U.S. Food and Drug Administration (FDA) — Approval of osimertinib for locally advanced, unresectable Stage III NSCLC (September 25, 2024): https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-osimertinib-locally-advanced-unresectable-stage-iii-non-small-cell-lung-cancer
-
U.S. Food and Drug Administration (FDA) — Approval of osimertinib with chemotherapy for EGFR-mutated NSCLC (February 16, 2024): https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-osimertinib-chemotherapy-egfr-mutated-non-small-cell-lung-cancer
-
AstraZeneca Press Release — Tagrisso with chemotherapy approved in the US for EGFR-mutated advanced lung cancer (February 2024): https://www.astrazeneca-us.com/media/press-releases/2024/tagrisso-osimertinib-with-the-addition-of-chemotherapy-approved-in-the-us-for-patients-with-egfr-mutated-advanced-lung-cancer.html
-
AstraZeneca Press Release — Tagrisso granted Priority Review in the US for unresectable Stage III EGFR-mutated lung cancer (June 2024): https://www.astrazeneca-us.com/media/press-releases/2024/tagrisso-osimertinib-granted-priority-review-in-the-us-for-patients-with-unresectable-stage-iii-egfr-mutated-lung-cancer.html
-
Lu S, Kato T, Dong X, et al. — Osimertinib after chemoradiotherapy in Stage III EGFR-mutated NSCLC (LAURA Trial), New England Journal of Medicine, 2024. doi:10.1056/NEJMoa2402614
-
Oncology News Central — Treatment Guide for Osimertinib (Tagrisso), including NCCN December 2025 guidelines update: https://www.oncologynewscentral.com/drugs/guide/osimertinib-nsclc
-
AJMC (American Journal of Managed Care) — FDA Approves Tagrisso for Adjuvant Treatment of Early-Stage EGFR-Mutated NSCLC (ADAURA Trial, 2021): https://www.ajmc.com/view/fda-approves-tagrisso-for-adjuvant-treatment-of-early-stage-egfr-mutated-nsclc
-
Cancer Network — FDA Approves Osimertinib/Chemo in EGFR-Mutant Lung Cancer (FLAURA2): https://www.cancernetwork.com/view/fda-approves-osimertinib-chemo-in-egfr-mutated-nsclc
-
Targeted Oncology — FDA OKs Osimertinib Plus Chemo in EGFR-Mutant Lung Cancer: https://www.targetedonc.com/view/fda-oks-osimertinib-plus-chemo-in-egfr-mutant-lung-cancer
-
American Cancer Society — Screening for Lung Cancer: 2023 Guideline Update (Wolf AMD et al.), CA: A Cancer Journal for Clinicians, 2024; 74(1): 50–81. doi:10.3322/caac.21811: https://acsjournals.onlinelibrary.wiley.com/doi/full/10.3322/caac.21811
-
American Cancer Society — New Lung Cancer Screening Guidelines, cancer.org: https://www.cancer.org/research/acs-research-news/new-lung-cancer-screening-guidelines-urge-more-to-get-ldct.html
-
PubMed / National Library of Medicine — Screening for Lung Cancer: 2023 ACS guideline update: https://pubmed.ncbi.nlm.nih.gov/37909877/
-
Nierengarten MB — Updated American Cancer Society Lung Cancer Screening Guidelines, Cancer, 2024; 130(5): 656–657. doi:10.1002/cncr.35223: https://acsjournals.onlinelibrary.wiley.com/doi/full/10.1002/cncr.35223
-
Pharmaceutical Commerce — FDA Approves Tagrisso With Chemotherapy for EGFR-Mutated Advanced Lung Cancer: https://www.pharmaceuticalcommerce.com/view/fda-approves-tagrisso-with-chemotherapy-for-egfr-mutated-advanced-lung-cancer
Medical Disclaimer
This article is provided strictly for educational, informational, and awareness purposes only. The real-life patient stories and examples shared in this article are drawn from clinical experiences, patient interactions, and commonly observed scenarios in oncology practice. They are presented for educational and awareness purposes only. Names and identifying details have been changed to protect patient privacy. While these accounts reflect authentic experiences encountered in medical practice, they have not been independently documented or published by us as formal case studies. Individual experiences with cancer diagnosis, treatment, and outcomes vary widely, and no single story should be taken as representative of what any reader may experience. It is not intended to be, and should not be construed as, professional medical advice, diagnosis, treatment, or a substitute for consultation with qualified healthcare professionals.
No Doctor-Patient Relationship — The information presented in this article does not establish a doctor-patient relationship between the reader and the author, publisher, or any affiliated entities. No medical decisions should be made based solely on the content of this article.
Consult Qualified Medical Professionals — If you are experiencing any symptoms mentioned in this article, have been diagnosed with cancer, or have concerns about cancer risk, seek immediate consultation with qualified oncologists, physicians, or appropriate medical specialists. For medical emergencies, contact emergency services immediately.
Individual Medical Situations Vary — Every person's medical condition, health history, risk factors, cancer type, and circumstances are unique. Diagnostic tests, treatment options, and medical recommendations must be tailored to individual patients through direct consultation with licensed healthcare providers who have access to complete medical histories and can perform proper clinical evaluations.
Not a Recommendation for Specific Tests or Treatments — References to biopsies, imaging studies, blood tests, tumor markers, chemotherapy, radiation therapy, immunotherapy, targeted therapy, surgery, or any other diagnostic procedures and treatments in this article are for informational purposes only and do not constitute recommendations that you should or should not undergo these tests or treatments. All decisions regarding medical testing, diagnosis, and treatment should be made in consultation with qualified healthcare professionals based on your specific medical situation.
No Guarantee of Accuracy or Completeness — While efforts have been made to provide accurate information, medical knowledge continuously evolves, particularly in the rapidly advancing field of oncology. The information in this article may not reflect the most current research, clinical guidelines, treatment protocols, or medical practices. The author and publisher make no representations or warranties regarding the accuracy, completeness, or timeliness of the content.
Do Not Disregard or Delay Professional Medical Advice — Never disregard, avoid, or delay obtaining professional medical advice from qualified healthcare providers because of something you have read in this article. Early detection and prompt treatment significantly improve cancer outcomes.
Third-Party Resources and Links — Any references to third-party medical services, clinics, doctors, cancer centers, or external websites are provided for informational purposes only and do not constitute endorsements.
Limitation of Liability — To the fullest extent permitted by law, the author, publisher, and affiliated entities disclaim all liability for any direct, indirect, incidental, consequential, or punitive damages arising from the use of, or reliance on, information contained in this article.
Geographic and Regulatory Considerations — Medical regulations, standards of care, insurance coverage, availability of diagnostic tests, treatment protocols, and access to cancer therapies vary by country, region, and healthcare system.
Clinical Trials and Experimental Treatments — Any references to clinical trials or investigational therapies are for informational purposes only. Participation in clinical trials should only be considered after thorough discussion with your oncology team.
Your Responsibility — You acknowledge that you are solely responsible for your own health decisions and that you will consult with appropriate licensed healthcare professionals before making any medical decisions or undergoing any diagnostic tests or treatments.
Acknowledgment — By reading and using the information in this article, you acknowledge that you have read, understood, and agreed to this disclaimer in its entirety. You further acknowledge that this article has been created with the assistance of artificial intelligence. While every effort has been made to ensure accuracy, AI-generated content may occasionally contain errors, omissions, or inaccuracies. The information presented here is intended solely for educational and informational purposes and should not be relied upon as a substitute for professional medical advice. Readers are strongly encouraged to consult qualified healthcare professionals, refer to peer-reviewed medical literature, and cross-reference information from established clinical sources before making any health-related decisions.
Last Updated: 20th February 2026
What's Your Reaction?
 Like
0
Like
0
 Dislike
0
Dislike
0
 Love
0
Love
0
 Funny
0
Funny
0
 Angry
0
Angry
0
 Sad
0
Sad
0
 Wow
0
Wow
0