When Prostate Cancer Stops Responding to Hormone Therapy: What Every Man Needs to Know About Castration-Resistant Prostate Cancer
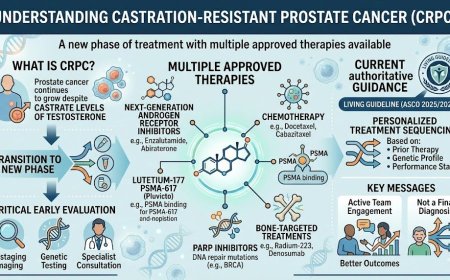
Castration-resistant prostate cancer (CRPC) occurs when prostate cancer continues to grow despite castrate levels of testosterone. It is not a final diagnosis but a new phase of treatment with multiple approved therapies available — including next-generation androgen receptor inhibitors, chemotherapy, lutetium-177 PSMA-617 (Pluvicto), PARP inhibitors for patients with DNA repair gene mutations, and bone-targeted treatments. Early comprehensive evaluation — including restaging imaging, genetic testing, and specialist consultation — is essential. The ASCO 2025/2026 Living Guideline, the most current authoritative guidance on mCRPC, emphasizes personalized treatment sequencing based on prior therapy, genetic profile, and performance status. Men who engage actively with their oncology team at CRPC diagnosis consistently achieve better outcomes.
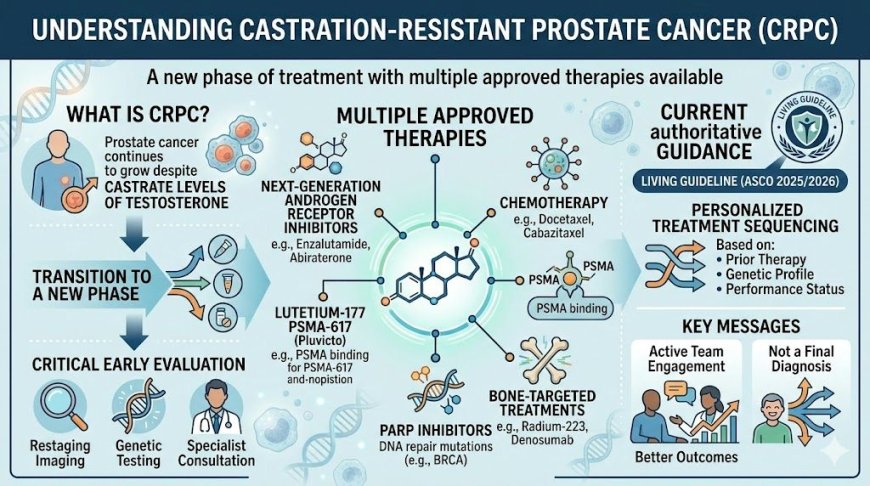
At A Glance
You were managing your prostate cancer. Hormone therapy was working. Then your doctor said three words that changed everything: "PSA is rising." You're confused, maybe scared. You're thinking: "Does this mean the treatment failed? Does this mean it's over?" It doesn't. Castration-resistant prostate cancer is a serious development — but it is not a death sentence. Modern oncology has transformed CRPC management dramatically in the last decade. What you do next, and how quickly you act, determines what happens in the chapters ahead. This article explains what CRPC is, why it happens, and what your real options are today.
The Moment Your Doctor Said "The Hormone Therapy Isn't Working Anymore"
You've been on androgen deprivation therapy — injections every few months, possibly tablets — and for a while, it was working. Your PSA was low. You were living your life. Going to work. Attending family functions. Managing.
Then came the blood test. Then the phone call. Or the face across the desk.
Your PSA — the prostate-specific antigen — has started rising again. Despite treatment. Despite testosterone being suppressed to castrate levels in your blood.
"Castration-resistant."
That word sounds frightening. Final. Like a door slamming shut.
Here's what it actually means: the cancer has found a way to keep growing even without the testosterone it used to feed on. It has adapted. That's concerning — but it's also something oncologists have learned to address with multiple newer, highly effective therapies that didn't exist even ten years ago.
Sound familiar? Then keep reading. Because what comes next matters enormously.
What Is Castration-Resistant Prostate Cancer — And Why Does It Happen?
Understanding Where It Begins
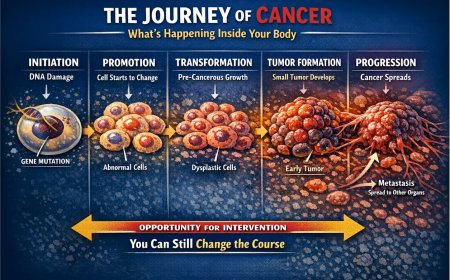
Prostate cancer is largely driven by male hormones called androgens — primarily testosterone. The most effective initial treatment for advanced prostate cancer is androgen deprivation therapy (ADT), which reduces testosterone to very low levels, starving the cancer cells of their fuel. This works well, often for months to years.
But prostate cancer cells are adaptive. According to multiple peer-reviewed studies published in journals including Translational Andrology and Urology and Cancers, the majority of patients with advanced or metastatic disease will progress to castration-resistant prostate cancer within two to three years of initiating ADT. This is not a treatment failure — it is a predictable biological evolution of the disease, and one that medicine has developed targeted strategies to address.
What "Castration-Resistant" Actually Means
The term refers to testosterone levels — not to treatment options. "Castration level" of testosterone means the blood testosterone level is extremely low (below 50 ng/dL), whether achieved through surgical removal of the testes (orchiectomy) or through medications (LHRH agonists or antagonists like leuprolide, goserelin, or degarelix). When cancer progresses despite these low testosterone levels, it is defined as castration-resistant.
Importantly, CRPC is not the end of treatment. It is a new phase of treatment — one that now has an expanding toolkit of approved therapies backed by strong clinical trial evidence.
The Biology Behind the Resistance
Cancer cells develop resistance through several mechanisms — including amplification of the androgen receptor, mutations in the receptor that allow it to be activated by other molecules, intratumoral androgen biosynthesis from the adrenal glands, and alternative signaling pathways that bypass the need for circulating testosterone entirely. Understanding this biology has directly led to the development of newer hormonal agents that work differently and more powerfully than earlier therapies.
The Two Categories of CRPC: What Stage Are You At?
Non-Metastatic CRPC (nmCRPC)
In this category, your PSA is rising despite castrate testosterone levels, but imaging studies show no detectable spread to other organs or bones. This can feel like a grey zone — something is clearly changing, but it hasn't visibly spread yet.
This is an important window. Modern next-generation androgen receptor inhibitors — apalutamide, enzalutamide, and darolutamide — have shown in large Phase III clinical trials (SPARTAN, PROSPER, and ARAMIS respectively, published in the New England Journal of Medicine) to significantly delay the development of metastases and improve overall survival in nmCRPC patients. These are approved therapies, not experimental, and they represent a genuine opportunity to slow or halt progression.
Metastatic CRPC (mCRPC)
In this category, cancer has spread beyond the prostate — most commonly to bones and lymph nodes, and sometimes to other organs. This is a more advanced stage, and it requires more active, multi-pronged treatment. However, even here, multiple approved therapies have extended survival and maintained quality of life in ways that were not possible a decade ago.
The distinction between nmCRPC and mCRPC matters because treatment approaches differ, and knowing exactly which category you're in through proper imaging is critical.
What You're Telling Yourself vs. What's Actually Happening
"My doctor said my PSA is rising but I feel fine — so it can't be that serious."
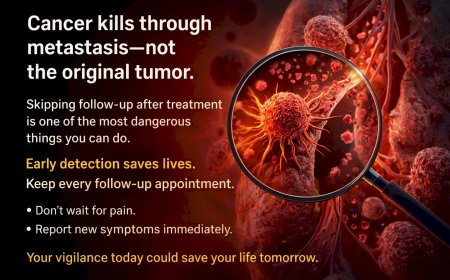
PSA is a biochemical marker. It can rise weeks or months before symptoms appear, and often does. Feeling well right now is good. But using it as a reason to delay or defer next steps in your treatment is a dangerous assumption. The value of acting at PSA progression — before you feel unwell — is precisely that you can start more effective therapies while your body is still strong enough to tolerate and benefit from them.
"Hormone therapy stopped working, so maybe nothing will work."
This is the fear talking, not the evidence. The treatment landscape for CRPC has undergone a revolution. Enzalutamide, abiraterone acetate, darolutamide, apalutamide, docetaxel chemotherapy, cabazitaxel, radium-223 for bone metastases, lutetium-177 PSMA-617 (Pluvicto), and PARP inhibitors for specific genetic mutations are all approved, available therapies. Your oncologist now has genuine options — not last resorts, but evidence-based strategies.
"We'll just wait and see."
In oncology, watchful waiting has a defined role. But passively "waiting and seeing" after confirmed CRPC without a clear monitoring plan and a discussed treatment strategy is not the same thing. Ensure that "watch and wait" is a deliberate, informed clinical decision — not a default response to uncertainty.
The Treatment Landscape: What Is Available Today
Next-Generation Hormone Therapies
These medications work differently from older ADT. They block the androgen receptor more completely or block the production of androgens from sources other than the testes — including the adrenal glands and tumor tissue itself.
Abiraterone acetate (Zytiga), taken with prednisone, blocks adrenal androgen synthesis. It is approved for both nmCRPC and mCRPC and has demonstrated significant survival benefit in multiple large trials including COU-AA-301 and COU-AA-302, published in the New England Journal of Medicine.
Enzalutamide (Xtandi) blocks the androgen receptor directly and prevents it from functioning even when activated. It is approved across multiple CRPC settings and is among the most widely used agents in this disease.
Darolutamide (Nubeqa) and apalutamide (Erleada) are newer agents in the same class, also approved with strong efficacy data across several settings.
According to the 2025/2026 ASCO Living Guideline on Systemic Therapy in Metastatic Castration-Resistant Prostate Cancer — ASCO's first continuously updated guideline in genitourinary oncology — prior systemic therapy determines subsequent treatment selection, and ADT should be continued indefinitely alongside these agents.
Chemotherapy
Docetaxel remains an important treatment in mCRPC, particularly in patients with rapidly progressing disease or those who are fit enough to tolerate chemotherapy. Cabazitaxel is available for patients whose disease has progressed after docetaxel, with both agents having demonstrated survival benefit in Phase III trials.
Lutetium-177 PSMA-617 (Pluvicto) — Radioligand Therapy
This represents one of the most significant advances in CRPC treatment in recent years. Lutetium-177 PSMA-617 (Pluvicto) is a targeted radioligand therapy that delivers radiation directly to prostate-specific membrane antigen (PSMA)-expressing prostate cancer cells.
The FDA initially approved Pluvicto on March 23, 2022, based on data from the landmark Phase III VISION trial (NEJM, 2021), for patients with PSMA-positive mCRPC who had previously received both an androgen receptor pathway inhibitor and taxane-based chemotherapy. In March 2025, the FDA expanded this approval to include patients who have received ARPI therapy and are considered appropriate to delay taxane-based chemotherapy — an important earlier access point.
PSMA PET-CT imaging, which is now available at major oncology centers in India, is required to determine eligibility for this therapy. It is significantly more sensitive than conventional bone scan and CT imaging for detecting prostate cancer spread.
PARP Inhibitors for Specific Genetic Mutations
Genomic alterations in DNA damage response (DDR) genes — including BRCA2, BRCA1, ATM, CDK12, and CHEK2 — are present in 20–30% of mCRPC patients, according to multiple studies including a landmark analysis published in the New England Journal of Medicine by Pritchard et al. (2016) which evaluated 692 men with metastatic prostate cancer. These mutations make cancer cells particularly vulnerable to a class of drugs called PARP inhibitors.
For patients with BRCA1/2 alterations who did not receive prior androgen receptor pathway inhibitor (ARPI) therapy, the 2025 ASCO Living Guideline recommends the combination of a PARP inhibitor with an ARPI (talazoparib + enzalutamide, or olaparib/niraparib + abiraterone). For those with BRCA1/2 alterations who received prior ARPI, olaparib monotherapy showed overall survival benefit in the PROfound trial (NEJM, 2020).
Germline and somatic genetic testing is now a standard recommendation in mCRPC evaluation per ASCO, NCCN, and European Association of Urology (EAU) guidelines alike.
Bone-Targeted Therapies
Radium-223 (Xofigo) is a targeted radiopharmaceutical for patients with mCRPC that has spread to bones but not other organs, with demonstrated overall survival benefit in the Phase III ALSYMPCA trial (NEJM, 2013). Per the 2026.1 ASCO Living Guideline, radium-223 is recommended for patients with symptomatic bone-only disease.
The ASCO Living Guideline also strongly recommends bone-protective agents — denosumab (Prolia) or zoledronic acid (Reclast) — for mCRPC patients with bone metastases, including during radium-223 therapy, to prevent fractures and skeletal-related events. This recommendation is based partly on the EORTC 1333/PEACE-3 trial.
Immunotherapy
Sipuleucel-T (Provenge) is an FDA-approved cellular immunotherapy for mCRPC that activates the patient's own immune cells against prostate cancer. While not widely available in India currently, it is part of the approved treatment toolkit and may be accessible through medical tourism or clinical trial participation.
Clinical Trials
Clinical trials are not just a "last resort." They represent access to the most advanced therapies before mainstream approval. For CRPC specifically, there are active trials investigating novel androgen receptor inhibitors, lutetium-PSMA combinations, radiopharmaceuticals, and immunotherapy combinations. The 2025 ASCO Living Guideline specifically emphasized the importance of clinical trial participation in CRPC management.
A Real Story: Rajesh, 67, Mumbai
Rajesh was diagnosed with advanced prostate cancer at age 62. His PSA at diagnosis was elevated, and scans showed lymph node involvement. He started on LHRH agonist therapy, and for nearly three years, his PSA remained low and he lived normally — work, travel, family.
At his 35-month check-up, his PSA had risen to 8. By his next check-up, it was 14. His oncologist explained that his cancer had become castration-resistant.
Rajesh's first reaction was to stop coming for follow-ups. "I was terrified. I thought it meant the cancer had won and there was nothing left to try." His family eventually convinced him to return.
His oncologist ran comprehensive testing including genomic profiling. Rajesh was found to carry a BRCA2 mutation. He was started on enzalutamide first, and when his disease later progressed, he transitioned to a PARP inhibitor through a compassionate use program. Two years after his CRPC diagnosis, he remains active and engaged in his grandchildren's lives.
His reflection: "I wasted three months in fear. Those three months could have cost me the chance to start treatment while I was still strong. My oncologist had options. I just didn't know it."
(This is a composite clinical scenario drawn from common oncology practice. Name and identifying details are fictional and used for educational illustration.)
The ACS and Major Society Guidelines: What They Recommend
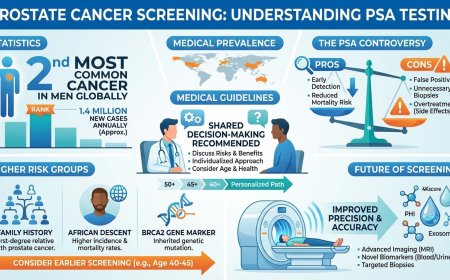
The American Cancer Society (ACS) recommends that asymptomatic men who have at least a 10-year life expectancy have an opportunity to make an informed decision with their healthcare provider about prostate cancer screening, including PSA testing. This decision-making process should begin at age 50 for men at average risk, and earlier for those at higher risk — including Black men and those with a first-degree relative diagnosed with prostate cancer before age 65. The ACS does not recommend routine, universal screening but strongly emphasizes informed, shared decision-making.
For men already diagnosed with prostate cancer who progress to CRPC, the following are consistent recommendations across major guidelines — ACS, ASCO (2025/2026 Living Guideline), NCCN, AUA/ASTRO/SUO, and EAU:
Continue ADT indefinitely after CRPC diagnosis. Begin early somatic and germline genetic testing. Use PSMA PET imaging for restaging. Administer bone-protective agents for patients with bone metastases. Sequence treatments based on prior therapy, genetic profile, performance status, and disease burden. Integrate early palliative care.
The Diagnostic Workup After CRPC Is Confirmed: What Your Doctor Should Be Doing
If you've been told your cancer is castration-resistant, the following evaluations are important components of a thorough workup. Discuss each of these with your oncologist:
Comprehensive restaging imaging — including bone scan and CT scan at minimum, and ideally PSMA PET-CT if available — to accurately determine whether and where the cancer has spread. PSMA PET imaging is significantly more sensitive than conventional scans and is now available in major oncology centers across India.
Germline and somatic genetic testing to screen for mutations in BRCA1, BRCA2, ATM, CDK12, CHEK2, and other DNA repair genes. This is now a strong recommendation in both ASCO and NCCN guidelines and directly guides treatment selection, including eligibility for PARP inhibitors and, in MSI-high cases, pembrolizumab.
Pathological reassessment in selected cases, as a small percentage of CRPC cases undergo transformation to neuroendocrine prostate cancer — a more aggressive variant that requires a different treatment approach. This should be discussed with your treating oncologist if you have rapidly progressing disease with minimal PSA rise.
Performance status assessment to determine fitness for chemotherapy and other intensive treatments, and to inform quality-of-life-centered decision making.
The Deadly Excuses That Delay Life-Saving Action
"I'll wait for my PSA to get higher before we talk about changing treatment."
There is no clinical benefit to waiting for disease to worsen before acting. Earlier intervention — while performance status is good and disease burden is lower — is associated with better outcomes across every major clinical trial in CRPC. The ASCO Living Guideline explicitly recommends early adoption of genetic testing and early integration of palliative care, not watchful delay.
"The treatments are too harsh — I don't want to feel sick all the time."
Modern CRPC therapies, particularly the oral next-generation hormone agents like enzalutamide, abiraterone, apalutamide, and darolutamide, are generally well tolerated in clinical trials. Side effects exist and should be discussed in full with your oncologist, but assuming that all cancer treatment means severe, constant illness is an outdated perception that prevents men from accessing treatments that could extend their lives with maintained quality.
"Private hospitals in India can't offer the same treatments as abroad."
Major cancer centers in India — Tata Memorial Hospital in Mumbai, AIIMS in Delhi, Apollo Cancer Centres, Fortis Cancer Institute, HCG Oncology, and others — have access to most approved CRPC therapies. India has also witnessed rapid adoption of PSMA PET imaging and genomic testing for prostate cancer. The key is finding a uro-oncologist or medical oncologist who manages a high volume of prostate cancer patients.
"My family doesn't need to know how serious this is."
CRPC management often involves complex decisions about treatment sequencing, quality of life trade-offs, and long-term planning. Having your family informed and involved — particularly for family members who may themselves carry inherited mutations (such as BRCA2, which is relevant for daughters and sisters) — is not a burden. It is a clinical and personal strength.
What You Must Do Right Now
Step 1: Confirm your exact diagnosis. Are you nmCRPC or mCRPC? Ask your doctor to clearly explain the staging and imaging findings in terms you understand.
Step 2: Ask specifically about genetic testing. If you have not had germline or somatic genomic testing, ask your oncologist why not. This is now a standard recommendation per ASCO's 2025/2026 Living Guideline, NCCN guidelines, and EAU guidelines.
Step 3: Request updated imaging if your last scan was more than three to four months ago. Restaging at CRPC diagnosis is critical — particularly PSMA PET-CT if available — because treatment decisions depend on accurate disease extent.
Step 4: Ask your oncologist directly: "What is the sequencing plan for my treatment, and what are the decision points?" Understanding the roadmap — not just the next step — helps you engage actively.
Step 5: Seek a second opinion if you have any doubt. In complex oncology cases, second opinions at high-volume cancer centers frequently lead to refined or revised treatment plans. It is your right and, in CRPC, often your advantage.
Professional Support Options
If you or a loved one has been diagnosed with castration-resistant prostate cancer, is seeking clarity on treatment options, or wants a second opinion from experienced oncologists, do not navigate this alone.
Connect with experienced U.S.-based cancer specialists for a comprehensive second-opinion consultation. They will carefully review your case and help determine the most appropriate next steps for your individual health needs:
👉 https://myamericandoctor.com/our-doctors/
You may also choose to enroll in our upcoming concierge medical clinic in India, Global Concierge Doctors. We offer U.S.-style primary care with 24/7 access to India-based physicians for ongoing guidance on any health concern. When required, we coordinate referrals to trusted specialists in India and the U.S. for advanced evaluation and care.
Your health decisions today shape your life tomorrow.
The Final Word
Castration-resistant prostate cancer is not a dead end. It is a turning point — one that demands a new strategy, not surrender. The men who do best are not those who were lucky enough to have an easy disease. They are the ones who stayed engaged, asked the right questions, demanded proper testing, and worked actively with their oncology team instead of waiting passively for instructions.
Your cancer adapted. Now it's your turn.
You have more options than you may realize. You have time — if you use it well. The decision you make in the next days and weeks about whether to pursue comprehensive evaluation, updated staging, genetic testing, and specialized treatment will shape what your next years look like.
Choose engagement over fear. Choose information over avoidance. Choose action over waiting.
Reader Poll
Have you or a loved one been told that hormone therapy is no longer controlling prostate cancer?
- Yes, and I'm actively pursuing next steps with my oncologist
- Yes, but I'm not sure what to do next
- No, but I want to understand CRPC better
- Currently in the process of getting a second opinion
Share This Article
Know a man managing prostate cancer who has been told his PSA is rising? Know a family navigating CRPC and unsure of what treatment options exist? Share this article. The information here could change what happens next for someone you love.
Last Updated: 27th February 2026
Sources
1. ASCO Living Guideline — Primary Guideline Source
Garje R, Riaz IB, Naqvi SAA, et al. Systemic Therapy in Patients With Metastatic Castration-Resistant Prostate Cancer: ASCO Living Guideline, Version 2026.1. Journal of Clinical Oncology. Published July 10, 2025. DOI: 10.1200/JCO-25-00007 https://ascopubs.org/doi/10.1200/JCO-25-00007
2. ASCO Living Guideline — Clinical Insights
Garje R, Riaz IB, Naqvi SAA, Rumble RB, Parikh RA. Systemic Therapy in Patients With Metastatic Castration-Resistant Prostate Cancer: ASCO Guideline Clinical Insights. JCO Oncology Practice. 2025. https://ascopubs.org/doi/10.1200/OP-25-00747
3. ASCO Post — Living Guideline Coverage
ASCO's First Living Guideline in GU Cancers Reflects Recent Practice-Changing Trials on Systemic Treatment of mCRPC. The ASCO Post. February 25, 2026. https://ascopost.com/issues/february-25-2026/
4. ACS Prostate Cancer Early Detection Guidelines
American Cancer Society. Recommendations for Prostate Cancer Early Detection. https://www.cancer.org/cancer/types/prostate-cancer/detection-diagnosis-staging/acs-recommendations.html
5. ACS Prevention and Early Detection Guidelines
American Cancer Society. Prevention and Early Detection Guidelines. https://www.cancer.org/health-care-professionals/american-cancer-society-prevention-early-detection-guidelines.html
6. AUA/SUO Early Detection Guideline
American Urological Association / Society of Urologic Oncology. Early Detection of Prostate Cancer: AUA/SUO Guideline (2026 Amendment). https://www.auanet.org/guidelines-and-quality/guidelines/early-detection-of-prostate-cancer-guideline
7. FDA Approval — Lutetium-177 PSMA-617 (Pluvicto) — Initial Approval
Fallah J, Agrawal S, Gittleman H, et al. FDA Approval Summary: Lutetium Lu 177 Vipivotide Tetraxetan for Patients with Metastatic Castration-Resistant Prostate Cancer. Clinical Cancer Research. May 1, 2023; 29(9): 1651–1657. https://aacrjournals.org/clincancerres/article/29/9/1651/725864/
8. FDA Expanded Approval — Lutetium-177 PSMA-617 (Pluvicto) — 2025
FDA Expands Pluvicto's Metastatic Castration-Resistant Prostate Cancer Indication. FDA.gov. March 28, 2025. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-expands-pluvictos-metastatic-castration-resistant-prostate-cancer-indication
9. VISION Trial — NEJM
Sartor O, de Bono J, Chi KN, et al. Lutetium-177–PSMA-617 for Metastatic Castration-Resistant Prostate Cancer. New England Journal of Medicine. 2021; 385(12): 1091–1103. https://www.nejm.org/doi/full/10.1056/NEJMoa2107322
10. PSMAfore Trial — Lancet
Morris MJ, Castellano D, Herrmann K, et al. 177Lu-PSMA-617 versus a change of androgen receptor pathway inhibitor therapy for taxane-naïve patients with progressive metastatic castration-resistant prostate cancer (PSMAfore). Lancet. 2024; 404(10459): 1227–1239. https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(24)01653-2/fulltext
11. PROfound Trial — PARP Inhibitor in mCRPC (BRCA mutations)
de Bono J, Mateo J, Fizazi K, et al. Olaparib for Metastatic Castration-Resistant Prostate Cancer. New England Journal of Medicine. 2020; 382(22): 2091–2102. https://www.nejm.org/doi/full/10.1056/NEJMoa1911440
12. Inherited DNA-Repair Gene Mutations in Metastatic Prostate Cancer
Pritchard CC, Mateo J, Walsh MF, et al. Inherited DNA-Repair Gene Mutations in Men with Metastatic Prostate Cancer. New England Journal of Medicine. 2016; 375(5): 443–453. https://www.nejm.org/doi/full/10.1056/NEJMoa1603144
13. DDR Gene Alterations in mCRPC — Frequency and Outcomes
Széles ÁD, Teutsch B, et al. Therapeutic sensitivity to standard treatments in BRCA positive metastatic castration-resistant prostate cancer patients — a systematic review and meta-analysis. Prostate Cancer and Prostatic Diseases. December 2022. https://www.nature.com/articles/s41391-022-00626-2
14. BRCA-Deficient mCRPC — Prognosis and Genomics
Fettke H, Dai C, Kwan EM, et al. BRCA-deficient metastatic prostate cancer has an adverse prognosis and distinct genomic phenotype. eBioMedicine (The Lancet). 2023; 95: 104738. https://www.thelancet.com/journals/ebiom/article/PIIS2352-3964(23)00303-1/fulltext
15. Abiraterone and Olaparib Combination in mCRPC
Clarke N, Wiechno P, Alekseev B, et al. Abiraterone and Olaparib for Metastatic Castration-Resistant Prostate Cancer. NEJM Evidence. 2022; EVIDoa2200043. https://evidence.nejm.org/doi/full/10.1056/EVIDoa2200043
16. Mechanisms of Resistance in CRPC
Chandrasekar T, Yang JC, Gao AC, Evans CP. Mechanisms of resistance in castration-resistant prostate cancer (CRPC). Translational Andrology and Urology. 2015; 4(3): 365–380. https://pmc.ncbi.nlm.nih.gov/articles/PMC4708226/
17. ADT Resistance Timeline
Wang Z, Yang X, Gao X, et al. Advances in the Current Understanding of the Mechanisms Governing the Acquisition of Castration-Resistant Prostate Cancer. Cancers. 2022; 14(15): 3744. https://www.mdpi.com/2072-6694/14/15/3744
18. Annals of Oncology — CAPTURE Study: HRR Mutations in mCRPC
Széles ÁD, et al. Treatment patterns and outcomes in metastatic castration-resistant prostate cancer patients with and without somatic or germline alterations in homologous recombination repair genes. Annals of Oncology. February 2024. https://www.annalsofoncology.org/article/S0923-7534(24)00043-7/fulltext
19. Baptist Health / Miami Cancer Institute — ASCO Guideline Commentary
Miami Cancer Institute Researchers Spearhead ASCO Guideline Update on Systemic Therapy for Metastatic Castration-Resistant Prostate Cancer. https://physicianresources.baptisthealth.net/news/miami-cancer-institute-researchers-spearhead-asco-guideline-update
20. Time to CRPC After ADT
Tamada S, et al. Time to progression to castration-resistant prostate cancer after commencing combined androgen blockade for advanced hormone-sensitive prostate cancer. PMC / PLoS ONE. 2018. https://pmc.ncbi.nlm.nih.gov/articles/PMC6319345/
Medical Disclaimer
This article is provided strictly for educational, informational, and awareness purposes only. The real-life patient stories and examples shared in this article are drawn from clinical experiences, patient interactions, and commonly observed scenarios in oncology practice. They are presented for educational and awareness purposes only. Names and identifying details have been changed or are fictional composites created to protect patient privacy. While these accounts reflect scenarios authentic to medical practice, they have not been independently documented or published by us as formal case studies. Individual experiences with cancer diagnosis, treatment, and outcomes vary widely, and no single story should be taken as representative of what any reader may experience. It is not intended to be, and should not be construed as, professional medical advice, diagnosis, treatment, or a substitute for consultation with qualified healthcare professionals.
No Doctor-Patient Relationship
The information presented in this article does not establish a doctor-patient relationship between the reader and the author, publisher, or any affiliated entities. No medical decisions should be made based solely on the content of this article.
Consult Qualified Medical Professionals
If you are experiencing any symptoms mentioned in this article, have been diagnosed with cancer, or have concerns about cancer risk, seek immediate consultation with qualified oncologists, physicians, or appropriate medical specialists. For medical emergencies, contact emergency services immediately.
Individual Medical Situations Vary
Every person's medical condition, health history, risk factors, cancer type, and circumstances are unique. Diagnostic tests, treatment options, and medical recommendations must be tailored to individual patients through direct consultation with licensed healthcare providers who have access to complete medical histories and can perform proper clinical evaluations.
Not a Recommendation for Specific Tests or Treatments
References to biopsies, imaging studies (CT scans, PET scans, MRI, PSMA PET), blood tests, tumor markers, chemotherapy, radiation therapy, immunotherapy, radioligand therapy, targeted therapy, surgery, or any other diagnostic procedures and treatments in this article are for informational purposes only and do not constitute recommendations that you should or should not undergo these tests or treatments. All decisions regarding medical testing, diagnosis, and treatment should be made in consultation with qualified healthcare professionals based on your specific medical situation.
No Guarantee of Accuracy or Completeness
While efforts have been made to provide accurate information, and sources have been referenced from peer-reviewed and authoritative publications, medical knowledge continuously evolves, particularly in the rapidly advancing field of oncology. The information in this article may not reflect the most current research, clinical guidelines, treatment protocols, or medical practices. The author and publisher make no representations or warranties regarding the accuracy, completeness, or timeliness of the content.
Do Not Disregard or Delay Professional Medical Advice
Never disregard, avoid, or delay obtaining professional medical advice from qualified healthcare providers because of something you have read in this article. If you have questions or concerns about information presented here, discuss them with your personal physician or oncologist. Early detection and prompt treatment significantly improve cancer outcomes.
Third-Party Resources and Links
Any references to third-party medical services, clinics, doctors, cancer centers, clinical trials, or external websites are provided for informational purposes only and do not constitute endorsements. The author and publisher are not responsible for the content, services, or practices of any third-party entities.
Limitation of Liability
To the fullest extent permitted by law, the author, publisher, and affiliated entities disclaim all liability for any direct, indirect, incidental, consequential, or punitive damages arising from the use of, or reliance on, information contained in this article. This includes, but is not limited to, medical complications, treatment decisions, financial losses, or any other adverse outcomes.
Geographic and Regulatory Considerations
Medical regulations, standards of care, insurance coverage, availability of diagnostic tests, treatment protocols, and access to cancer therapies vary by country, region, and healthcare system. In India, availability and approval status of therapies mentioned in this article may differ from U.S. FDA approvals. Readers in India should consult with oncologists familiar with Indian regulatory approvals, drug availability, and institutional protocols. Information regarding procedures and medical practices may not be applicable to all geographic locations or healthcare settings.
Clinical Trials and Experimental Treatments
Any references to clinical trials, experimental treatments, or investigational therapies are for informational purposes only. Participation in clinical trials should only be considered after thorough discussion with your oncology team and a full understanding of all risks and benefits.
Your Responsibility
You acknowledge that you are solely responsible for your own health decisions and that you will consult with appropriate licensed healthcare professionals before making any medical decisions or undergoing any diagnostic tests or treatments.
Acknowledgment
By reading and using the information in this article, you acknowledge that you have read, understood, and agreed to this disclaimer in its entirety. You further acknowledge that this article has been created with the assistance of artificial intelligence. While every effort has been made to ensure accuracy and sources have been referenced from authoritative publications, AI-generated content may occasionally contain errors, omissions, or inaccuracies. The information presented here is intended solely for educational and informational purposes and should not be relied upon as a substitute for professional medical advice. Readers are strongly encouraged to consult qualified healthcare professionals, refer to peer-reviewed medical literature, and cross-reference information from established clinical sources before making any health-related decisions.
What's Your Reaction?
 Like
0
Like
0
 Dislike
0
Dislike
0
 Love
0
Love
0
 Funny
0
Funny
0
 Angry
0
Angry
0
 Sad
0
Sad
0
 Wow
0
Wow
0